


Toxta 100 Units
$64
Comes with 1 saline solution (180mg/20mL)
Box: 100 units * 1 Vial
Manufacturer: JETEMA Co., South Korea
In stock
To change a warehouse go to "Shop" page
Approximate delivery: Tuesday, June 16
Pay by Card, Easily
Seamless checkout with major cards
Worry-Free Delivery
Any customs issue, we’ll reship or refund quickly
FedEx You Can Trust
Fast, trackable worldwide delivery
Application area Toxta 100 units
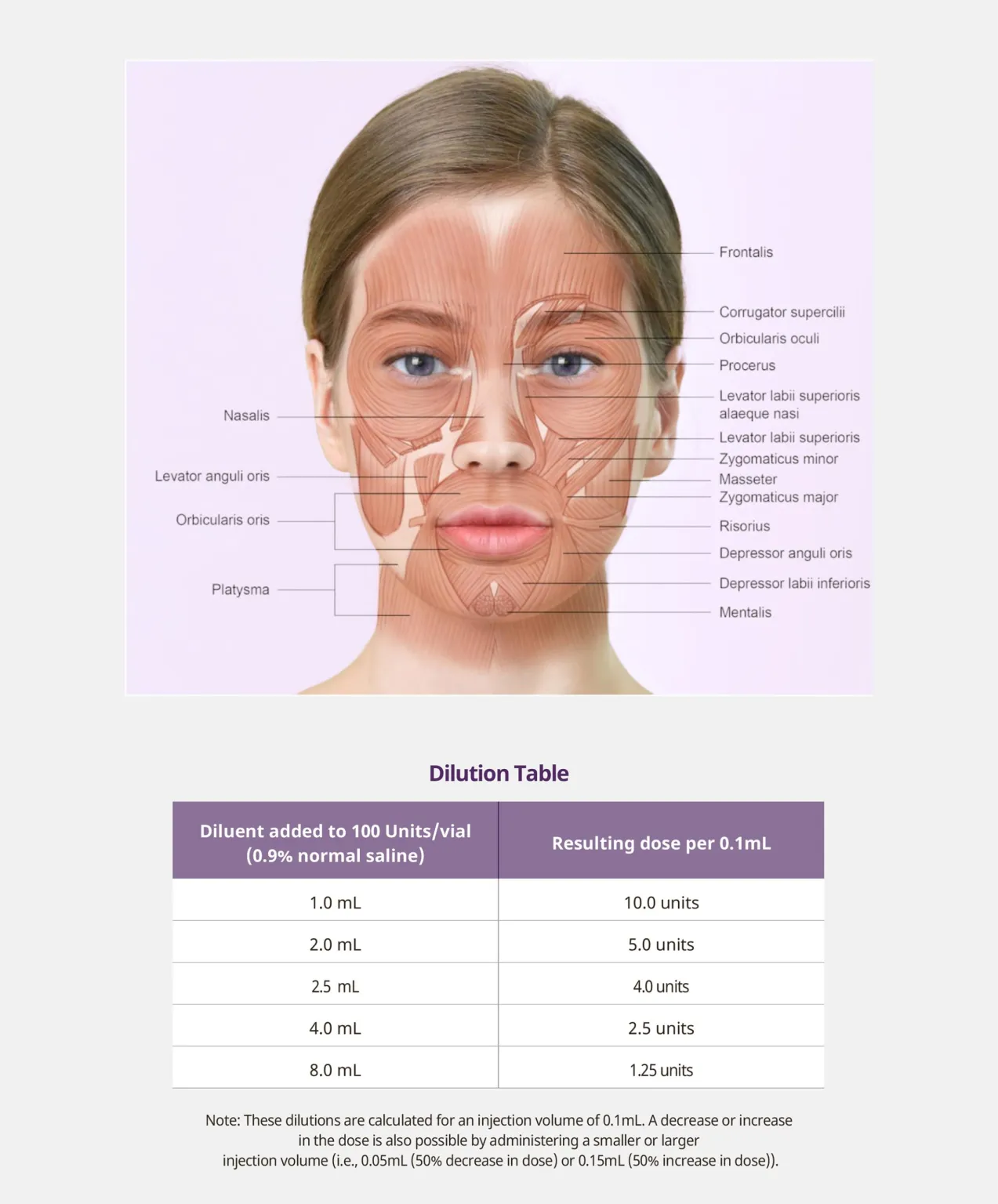
Intended for temporary improvement of dynamic glabellar lines in adults, this botulinum type A neurotoxin is used in targeted injections to relax the corrugator and procerus muscles. In standard protocols the single-use vial of Toxta 100 unit is reconstituted with 2.5 mL of 0.9% saline, yielding 4 U per 0.1 mL; a typical treatment for the glabella uses 20 U divided across five points (0.1 mL each) to achieve controlled relaxation and wrinkle reduction while preserving facial expression.
Storage is 2 – 8 °C, and the labeled shelf life is 36 months. The strain used is registered in GenBank (NIH/NCBI, CP046450), and handling follows single-vial aseptic technique to support reproducible results and a measured approach to aesthetic rejuvenation without systemic spread beyond the intended area.
Strengths of Toxta 100 units
Manufacturing emphasizes high purity and low accessory proteins: internal analytics report >99% purity and ~2.82 ng of neurotoxin protein per 100 U vial, factors associated with a lower theoretical risk of antibody formation compared with products containing more denatured proteins. Vacuum-drying technology, exclusion of animal-derived ingredients (and common plant allergens), plus a clear dilution table support dose accuracy and consistent effectiveness. In pre-clinical work and an adult Phase I comparison, safety was similar to a reference product with low rates of mild, transient events.
For clinicians evaluating Toxta botox alongside other botulinum toxins, the following technical attributes are frequently highlighted:
- High purity with documented strain sequence (GenBank CP046450)
- Reconstitution guidance that maps units per 0.1 mL (e.g., 4 U at 2.5 mL)
- Single-use vial, refrigerated storage at 2 – 8 °C, 36-month expiry
- Process design excluding animal components and certain allergenic plants
- Clinical and laboratory evaluations indicating non-inferior safety profiles
Use must comply with local regulations (e.g., FDA rules in the U.S.), and procedures should be performed by appropriately certified practitioners using established protocols.
Who is This Product For
Best suited to medical professionals trained in neurotoxin techniques who require a well-characterized product for precise muscle relaxant dosing, this formulation serves patients bothered by frown lines between the brows and some related fine lines where dynamic activity predominates.
Ideal candidates are adults seeking a conservative, localized reduction in movement with a predictable onset and duration, and who can adhere to aftercare and follow-up. It is not intended for individuals with hypersensitivity to toxin components, active infection at the injection site, or relevant neuromuscular disorders; patient selection should consider medical history, concomitant medicines, and risk tolerance. In clinical practice, documentation of baseline photographs, clear goals for appearance change, and informed consent are essential; outcome assessment should track symmetry, dose per point, and any adverse events to ensure adjustments at subsequent visits. While broader claims should be avoided, a cautious, protocol-driven approach helps align expectations about the scope of rejuvenation that can be achieved with Toxta botox-class agents.
Reviews
- Good for affordable priceGreat price-quality ratioPretty nice. I feel nabota is better. Especially when it comes to how fast product starts working
Only logged in customers who have purchased this product may leave a review.









